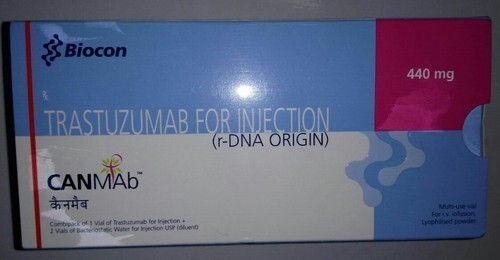
transtuzumab medicine
10 USD ($)/Piece
Product Details:
- Indication Breast Cancer, Gastric Cancer
- Dosage Form Injection
- Salt Composition Trastuzumab
- Enzyme Types Monoclonal Antibody
- Feature Highly purified, targeted anti-cancer therapy
- Ingredients Trastuzumab (Recombinant Humanized Monoclonal Antibody)
- Application Oncology, HER2-Positive Cancers
- Click to View more
X
transtuzumab medicine Price And Quantity
- 10 USD ($)/Piece
- 2.20 - 25.00 USD ($)/Piece
- 100 Piece
- Glass vials with rubber stoppers
- Prescription required
- Reconstitute with provided sterile water for injection
- Adults with HER2-positive cancers
- Antineoplastic Agent, HER2 Inhibitor
- Single dose vial
- cGMP compliant
- Non-animal origin; humanized antibody
- Use under medical supervision; for hospital use only
- Intravenous (IV) infusion
- 150 mg/vial
- Preservative free
- Compatible with 0.9% Sodium Chloride IV solution
transtuzumab medicine Product Specifications
- Oncology, HER2-Positive Cancers
- Monoclonal Antibody
- Breast Cancer, Gastric Cancer
- Approximately 6.0
- Injection
- Store at 2C 8C. Do not freeze.
- Trastuzumab
- Highly purified, targeted anti-cancer therapy
- Trastuzumab (Recombinant Humanized Monoclonal Antibody)
- Not Applicable (Monoclonal Antibody)
- Odorless
- 24 months
- Clear to slightly opalescent, colorless to pale yellow liquid
- Glass vials with rubber stoppers
- Prescription required
- Reconstitute with provided sterile water for injection
- Adults with HER2-positive cancers
- Antineoplastic Agent, HER2 Inhibitor
- Single dose vial
- cGMP compliant
- Non-animal origin; humanized antibody
- Use under medical supervision; for hospital use only
- Intravenous (IV) infusion
- 150 mg/vial
- Preservative free
- Compatible with 0.9% Sodium Chloride IV solution
transtuzumab medicine Trade Information
- mumbai or DHL or aramex or EMS
- Paypal, Western Union, Cash Advance (CA)
- 100 Piece Per Day
- 7 Days
- Contact us for information regarding our sample policy
- standard
- Australia, Central America, North America, South America, Eastern Europe, Western Europe, Middle East, Asia, Africa
- All India
- depend
Product Description
Canmab 440mg Injection is used in the treatment of breast and stomach cancer. It works by killing the cancer cells by inhibiting HER2 (human epidermal growth factor receptor protein).How Does Trastuzumab Work?
Trastuzumab acts by selectively binding to the HER2 protein found on certain cancer cells, inhibiting cell growth and proliferation. This targeted approach makes it highly effective in treating HER2-positive breast and gastric cancers, resulting in improved patient outcomes and reduced impact on healthy tissue.
Storage and Handling
This medicine should be stored in a refrigerator at 2C to 8C, avoiding freezing. It is supplied in glass vials with rubber stoppers and must be handled using aseptic techniques in hospital settings. Each vial is for single use, maintaining product sterility and efficacy.
Preparation and Dosage Administration Process
To prepare the dose, reconstitute the lyophilized powder using the provided sterile water for injection. The solution should be compatible with 0.9% sodium chloride IV solution for infusion. Administration must be under strict medical supervision, conforming to prescribed protocols and dosage guidelines.
FAQ's of transtuzumab medicine:
Q: How should Trastuzumab 150 mg/vial be administered for optimal efficacy?
A: Trastuzumab should be reconstituted with the provided sterile water for injection, then diluted with 0.9% sodium chloride IV solution before intravenous infusion. It must be administered by healthcare professionals in hospital settings, following prescribed protocols.Q: What are the main indications for using this medication?
A: Trastuzumab is indicated for the treatment of HER2-positive cancers, particularly in adult patients diagnosed with breast cancer and gastric cancer. Use requires positive confirmation of HER2 overexpression in tumors.Q: When should Trastuzumab therapy be started for patients?
A: Therapy should begin as soon as HER2-positivity is confirmed by a medical professional. The timing and dosing schedule depend on individual patient assessments and clinical protocols, always under medical supervision.Q: Where should Trastuzumab be stored prior to use?
A: The vials must be stored in a refrigerator at 2C to 8C. They should never be frozen and should be kept in their original glass containers until reconstitution and administration.Q: What process is involved in reconstituting and preparing this medicine for infusion?
A: Reconstitution requires aseptically adding sterile water for injection to the vial, gently swirling to dissolve the contents. The reconstituted solution is then further diluted in 0.9% sodium chloride IV solution for intravenous delivery.Q: What are the benefits of using a humanized monoclonal antibody like Trastuzumab in cancer therapy?
A: Trastuzumab offers a targeted approach, minimizing damage to healthy cells and improving effectiveness in eradicating HER2-positive cancer cells. This reduces side effects compared to non-targeted chemotherapy agents.Q: Is this product suitable for individuals with animal allergies?
A: Yes, Trastuzumab is produced using non-animal origin materials and is a recombinant humanized monoclonal antibody, making it appropriate for patients with animal-derived protein sensitivities.Tell us about your requirement

Price:
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
Mobile number
Email



 Send Inquiry
Send Inquiry Send SMS
Send SMS
